Studies
of the molecular basis of Drosophila melanogaster mechanosensation. Mechanosensory transduction underlies a wide range of senses including proprioception, touch, balance, and hearing. The pivotal element of the mechano-senses is a mechanically gated ion channel that transduces sound, pressure, or movement into changes in excitability of specialized sensory cells. Despite the prevalence of mechanosensory systems, little was known about the molecular nature of these mechanically gated ion channels. To identify components of mechanotransduction, Drosophila mechanosensory mutants were analyzed for defects in sensory physiology defined by electrophysiological recordings from mechanosensory bristles. My doctoral dissertation focused on the characterization of three of these mutants (nompC, nompH, and nompF) and demonstrated that not only was this genetic strategy ideal for mechanotransduction studies, it was also well suited for exploring the formation and function of sensory structures. Loss-of-function mutations in the no mechanoreceptor potential C (nompC) 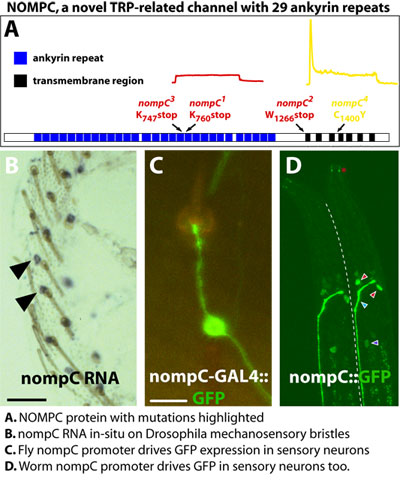 gene virtually abolished mechanosensory signaling while one causes profound
defects in adaptation. NompC encodes a novel ion channel essential
for mechanosensory transduction, making it the first identified eukaryotic
mechanosensitive channel. Containing 28 ankyrin repeats, NOMPC is a
unique member of the TRP family of cation channels. Drosophila
NOMPC and a C. elegans homologue are selectively expressed
in mechanosensory neurons. This work was published
in Science in 2000. Lastly, a vertebrate NOMPC was recently identified
by another group and shown to be expressed in mechanosensory tissue
including hair cells of the inner ear.
gene virtually abolished mechanosensory signaling while one causes profound
defects in adaptation. NompC encodes a novel ion channel essential
for mechanosensory transduction, making it the first identified eukaryotic
mechanosensitive channel. Containing 28 ankyrin repeats, NOMPC is a
unique member of the TRP family of cation channels. Drosophila
NOMPC and a C. elegans homologue are selectively expressed
in mechanosensory neurons. This work was published
in Science in 2000. Lastly, a vertebrate NOMPC was recently identified
by another group and shown to be expressed in mechanosensory tissue
including hair cells of the inner ear.Cytochrome P450s have generally been acknowledged as broadly tuned detoxifying enzymes. However, emerging evidence argues P450s 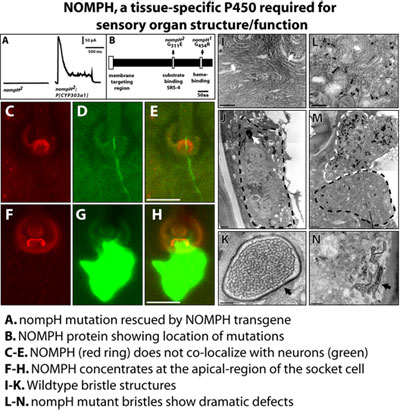 have
an integral role in cell signaling and developmental processes, via
their metabolism of retinoic acid, arachidonic acid, steroids, and other
cellular ligands. NompH, a mutant defective in the function
of peripheral chemo- and mechanoreceptor cells, encodes the cytochrome
P450 CYP303a1. CYP303a1 is essential for the development and structure
of external sensory organs which mediate the reception of vital mechanosensory
and chemosensory stimuli. Notably this P450 is expressed only in sensory
bristles, at the apical region of the socket cell. This exquisite tissue
and subcellular specificity illustrates an increasingly recognized aspect
of P450 function; namely, a strategy to process critical developmental
signals in a tissue- and cell-specific manner. This work was published
in Mechanisms of Development in 2004. have
an integral role in cell signaling and developmental processes, via
their metabolism of retinoic acid, arachidonic acid, steroids, and other
cellular ligands. NompH, a mutant defective in the function
of peripheral chemo- and mechanoreceptor cells, encodes the cytochrome
P450 CYP303a1. CYP303a1 is essential for the development and structure
of external sensory organs which mediate the reception of vital mechanosensory
and chemosensory stimuli. Notably this P450 is expressed only in sensory
bristles, at the apical region of the socket cell. This exquisite tissue
and subcellular specificity illustrates an increasingly recognized aspect
of P450 function; namely, a strategy to process critical developmental
signals in a tissue- and cell-specific manner. This work was published
in Mechanisms of Development in 2004.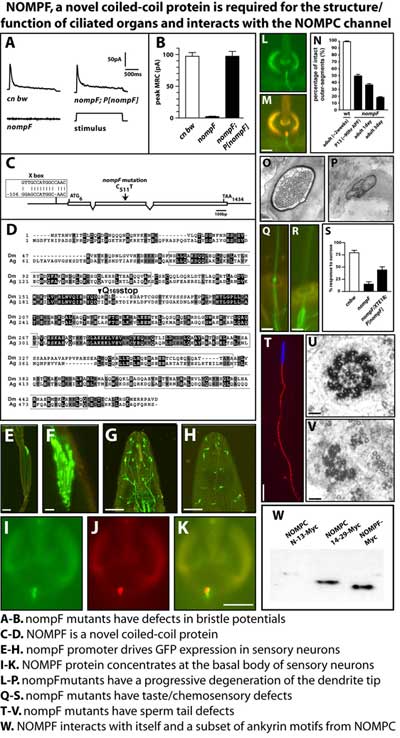
Cilia are central to the structural organization and function of sperm flagella and mechanosensory and chemosensory dendrites. An uncoordinated mutant, nompF, has dramatic defects in ciliated cells including degeneration of the outer segments of chemo- and mechanosensory sensory neurons which contain the transduction machinery. Additionally, nompF mutants have unstable axonemes which split and fragment, resulting in non-motile sperm. NOMPF, a novel coiled-coil protein, localizes to the base of the connecting cilium in neurons and to the axoneme in sperm tails. A demonstrated interaction with the mechanosensitive channel NOMPC together with nompF expression pattern and mutant phenotype suggests nompF has a critical role in assembly and maintenance of ciliated structures. |